- Digital Library
- AR-CAPS® Acid resistant capsules

Acid Resistant Capsules for Intestinal Delivery
Contact UsAR-CAPS®are produced from an exclusive formulation of Hypromellose and proprietary polymers, AR-CAPS® gastric acid-resistant hard capsules prevent the release of drugs in the stomach, making them a cost-effective, quality choice for a vast range of enteric delivery applications.
AR-CAPS® deliver medicine by disintegrating in the duodenum for intestinal absorption. With AR-CAPS®, costly and complex enteric coating processes are eliminated.
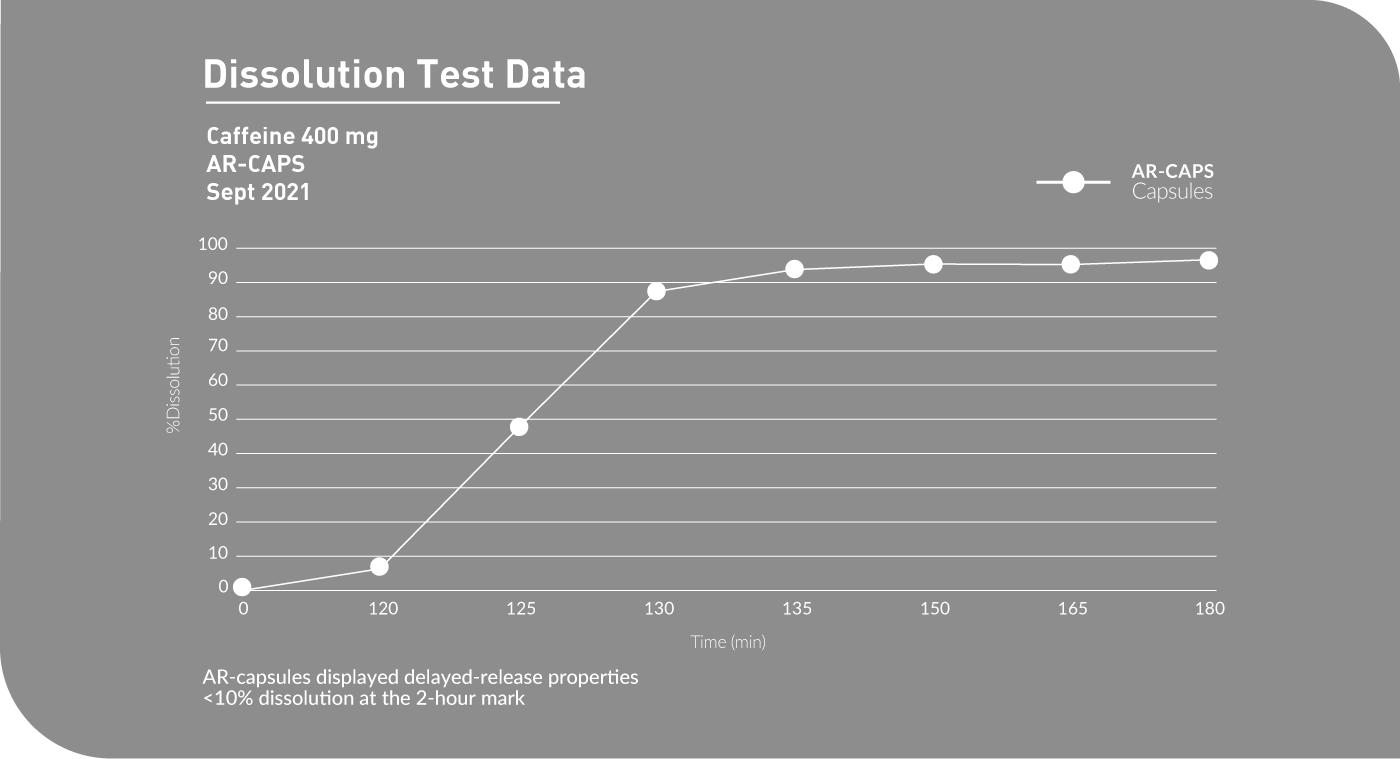
AR-CAPS® are naturally acid resistant, which eliminates the need for added chemicals, solvents or other coatings to delay the release of your formulation as it passes through the digestive tract. The capsule retains its stability against the pH levels of the stomach but breaks down when it reaches the higher pH levels of the small intestines. AR-CAPS® have a low moisture content (4%-10%), making them ideal for moisture sensitive or hygroscopic formulations.


AR-CAPS® are available in size 00, 0 and 1. Other sizes are available upon request.

Do you want to know more about AR-CAPS®? Contact us now!
- Less containment required when handling highly potent actives
- Better delivery of poorly soluble actives using lipidbased formulations and other liquid carriers
- Higher content uniformity of low-dose products
- Fewer excipients than tablets and powder-filled capsules
- Stronger abuse deterrence and tamper evidence
- Faster process development, scaleup and launch
- Unlimited combinations of printing schemes and colors
- GMP-compliant clinical batches
- Dedicated facility for all liquid fills
- Custom capsule and banding polymers
- Formulation and development services
- Full analytical lab
- DEA license for drug schedules I-V
- Blister and bottle packaging
- Stability and release testing
 style="margin: 0;"
style="margin: 0;" 